Aesthetic Procedures: Breast Augmentation with Implants

Table of Contents
- Breast augmentation with implants
- What incisions are used?
- Where is the implant-placed?
- What type of breast implants are used?
- Hybrid breast augmentation
- Which method is right for me?
- What can i expect after a breast augmentation
- Why choose polaris plastic surgery?
- Risks and complications
- Faqs
- References
.jpg?width=800&height=513&name=Incision%20points%20in%20breast%20implant-1%20(1).jpg)
Breast Augmentation With Implants
Breast augmentation with implants involves the placement of a breast implant in a suitable pocket underneath the breast, giving the breast more volume while minimizing the appearance of any scars. It is performed as day surgery under general anaesthesia, or local anaesthesia with sedation.
What incisions are used?
| Procedure | IMF approach | Periareolar approach incisions | Transaxillary approach |
|---|---|---|---|
| Advantages |
|
|
|
| Disadvantages |
|
|
|
For more information scroll right.
Where is the implant placed?
| Placement of implants | Subglandular plane | Subfascial plane | Under chest muscle |
|---|---|---|---|
| Position | Implants are placed under the breast and over the muscle directly under the breast tissue | Implants are placed under a fascial layer overlying the pectoralis major muscle | Implants are placed under the chest (pectoralis major) muscle |
| Advantages | The most natural plane in which to place the implant as it replaces the actual breast tissue | Chest muscle fascia acts as a barrier between the breast tissue and the implant | Provides robust coverage and reduced palpability of the implant |
| Disadvantages |
|
Possible risk of rippling deformity | Can lead to muscle weakness and potential animation deformity, where the implant can move when the chest muscle is activated |
| Recommended for | For patients with moderate to large breasts and/or thick chest skin | For patients with moderate to large breasts and/or thick chest skin | For patients who are thin with small breasts and thin chest skin |
For more information scroll right.
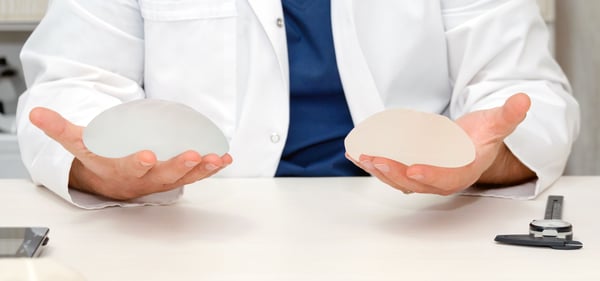
What type of breast implants are used?
Many breast implants used in modern day plastic surgery are silicone-filled. Compared to other fill materials such as saline, the latest silicone implants offer a more natural feel, last longer and are less prone to silicone gel bleed. Breast implants also come in different sizes, shapes, and surface linings.
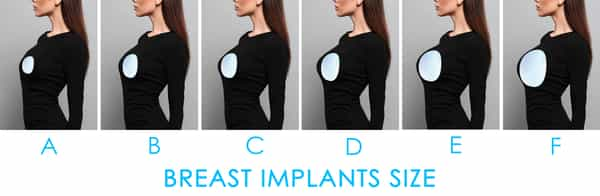
SIZE
A common query is how breast implant size relates to cup size. In general, about 150cc is required for each cup size increase. However, the eventual outcome is dependent on other factors as well including existing body shape and how much the breast and chest tissue can stretch. There is a natural limit to which the native skin can expand comfortably, and an overly large implant can appear unnatural.
SHAPE
In terms of shape, there are round- versus teardrop-shaped implants. Round implants have the advantage of not being subjected to malrotation, but does lead to a slightly more downward pointing nipple. Teardrop-shaped implants are also called anatomical implants, and help to simulate the natural breast shape with 45% of breast volume in the upper half and 55% of breast volume in the lower half. The leads to a less convex upper pole compared to the round implant, and a more forward or upward pointing nipple.
However, teardrop-shaped implants are susceptible to malrotation, and in most cases have a textured coating, which could increase the risk of breast implant associated anaplastic large cell lymphoma (BIA-ALCL).
EXTERNAL LINING
This brings us to the next big difference in breast implants, which is the external lining. Implants can be either smooth or textured. Smooth-surfaced implants have in the past been associated with higher rates of capsular contracture than textured implants, mainly in the subglandular plane. However, with better techniques, subfascial implant placement and increased understanding of the contributing factors to capsular contracture, this has become less of an issue.
Textured implants have rough surfaces created during the manufacturing process, which besides the afore-mentioned reduction in capsular contracture, are also purported to prevent implant malrotation. However, a recent development has been the discovery of BIA-ALCL, a form of lymphoma which has been primarily associated with textured implants. See our FAQ for information on BIA-ALCL.
In recent years, there has been the development of nano-textured implants, which is meant to bring the benefits of smooth and textured implants together in one package, whilst minimizing the risks of each. Our doctors are fully certified in the use of MotivaTM breast implants, which are nano-textured implants leading the way in terms of advanced design, natural feel and improved outcomes.

.png?width=307&height=437&name=New%20Project%20(1).png)
Dr. Adrian Ooi and Dr Pek Chong Han are certified Motiva™ users.
Hybrid breast augmentation
Which method is right for me?
Our surgeons will conduct a thorough discussion of your expectations and desires as well as the techniques available to help you decide on the most appropriate breast augmentation method for you to achieve a natural, pleasing look. The choice of incision, plane, and implant would be aimed at maximizing your appearance and outcome while minimizing any complications. Utilizing morphing software, they can also give you an idea of how different implants will suit your overall appearance.
What can I expect after a breast augmentation?
You may experience pain, swelling, and bruising for the first week, which is usually well controlled with medication. You will be reviewed in the clinic within the first 2 weeks, during which time you should take good care of your incisions and avoid any strenuous activity. As you progress, you can begin light exercise, and can usually resume full activity at 4 to 6 weeks after the procedure. Subsequently, you can wear a normal bra, and it would be best to avoid any heavy impact on the chest as it may lead to implant rupture.

Why choose Polaris Plastic Surgery?
Risks and complications
Rippling can occur early or late and is due to insufficient soft tissue coverage over the implant. Deformities and malposition can arise due to improper implant choice or placement, and require addressing in a secondary procedure.
Capsular contracture is the formation of a firm lining around the implant which may be painful, requiring implant removal or exchange, and happens in about 5% of cases. Very rarely, breast implant associated anaplastic large cell lymphoma (BIA-ALCL), a form of cancer, has been reported in relation to breast implants.
FAQs
What is breast implant associated anaplastic large cell lymphoma (BIA-ALCL)?
BIA-ALCL is a rare peripheral lymphoma which was first discovered in 1997. To date, more than 300 cases of BIA-ALCL have been reported in the United States alone, mainly occurring in patients who received textured-surface breast implants. The incidence of BIA-ALCL ranges from 1 case per 4,000 implants to 1 case per 30,000. As it is relatively new, knowledge on this topic continues to evolve. It most commonly presents as a late peri-implant collection more than one year following the breast implant surgery. Other less common symptoms include a breast mass, enlargement of the axillary lymph nodes, with fever and fatigue. The average time from implant placement to onset of BIA-ALCL is 10.7 years. BIA-ALCL may affect patients with either silicone- or saline-filled implants. BIA-ALCL may be diagnosed on ultrasound or MRI scan, in conjunction with the use of image-guided needle fluid aspiration or biopsy.
Most cases of BIA-ALCL are considered localized and have an early-onset form and can be cured by implant removal and complete excision of the implant capsule. Regular surveillance examinations imaging investigations may be required. Patients with more advanced disease may require chemotherapy and/or radiotherapy. If you already have breast implants, the US FDA states: “There is no need to change your routine medical care and follow-up”. Additional screening or removal of implants is not required for asymptomatic women. The FDA continues to affirm that “BIA-ALCL is a very rare condition”. Should you experience any of the symptoms described above, do arrange for a consultation with your plastic surgeon.
What is capsular contracture?
As a result of an inflammatory, foreign-body reaction, the body develops a capsule around any implant placed inside. This can sometimes be hastened by a low-level infective process. When severe enough, in breast implants this capsule can lead to capsular contracture. Capsular contracture itself has different grades of severity and can be classified into 4 stages (Baker’s classification):
Stage 1: Breast is soft, and implant is not palpable
Stage 2: Breast is solid, and implant is palpable but not visible
Stage 3: Breast is solid, and implant is palpable and visible
Stage 4: Breast is deformed and painful and implant is palpable and visible
How long do breast implants last?
Placed well with meticulous technique, breast implants can last a lifetime with low risks of capsular contracture. However, if this does happen, the usual timeframe for symptomatic capsular contracture is 10 years. This can be severe and distressful, and capsule removal or replacement with implants or fat grafts can be done as treatment.
What is the right age for a breast augmentation?
Breast augmentation can be done at any age once breasts are fully developed, which usually occurs by 18 years old. There is no age limit, though at a later age factors such medical suitability, skin quality, and breast ptosis must be considered.
Can I breastfeed after my breast augmentation?
Whether done using implants or fat grafting, breastfeeding is safe after breast augmentation.
1. Natrelle round silicone breast implants: Core Study results at 10 years. Spear SL, Murphy DK, Allergan Silicone Breast Implant U.S. Core Clinical Study Group. Plast Reconstr Surg. 2014 Jun; 133(6):1354-1361.
2. Hunstad JP, Webb LS. Subfascial breast augmentation: a comprehensive experience. Aesthetic Plast Surg. 2010 Jun;34(3):365-73
3. Wong CH, Samuel M, Tan BK, Song C. Capsular contracture in subglandular breast augmentation with textured versus smooth breast implants: a systematic review. Plast Reconstr Surg. 2006 Oct;118(5):1224-1236.
4. Maximiliano J, Munhoz AM, Pedron M, de Oliveira ACP, Duarte DW, Neto R, Portinho CP, Collares MVM. Hybrid Breast Augmentation: A Reliable Formula for Preoperative Assessment of Fat Graft Volume Based on Implant Volume and Projection. Aesthet Surg J. 2020 Jul 13;40(8):NP438-NP452.
5. Brown T. A Comprehensive Outcome Review of Subfascial Breast Augmentation over a 10-Year Period. Plast Reconstr Surg. 2020 Dec;146(6):1249-1257.
6. Doren EL, Miranda RN, Selber JC et al. U.S. epidemiology of breast implant-associated anaplastic large cell lymphoma. Plast Reconstr Surg 2017;139(5):1042-1050.
PATIENT STORIES
Post Massive Weight Loss Body Contouring
A 40-year-old lady consulted with Dr Pek with the desire to correct large amounts of excess skin and soft tissue. She had undergone Bariatric Surgery (Sleeve Gastrectomy) 2 years prior to her presentation and had successfully lost almost 30kg of weight. With ... Read more
Treat Gynecomastia with Minimally Invasive Methods in Singapore
A 38-year-old male engineer presented to Dr Pek with symptomatic growth of his chest/breast area. Since puberty, he complained of unusually prominent chest tissue around his nipple and areolar region. This had worsened gradually over the years, with the left ... Read more
Upper eyelid lift and eyebags rejuvenation
63 years old Chinese female with hypertension and hyperlipidemia presented to Dr Pek with significant droopiness of her upper eyelids on both sides, affecting the upper portion of her visual field. She would constantly feel that her upper eyelids were heavy, ... Read more
Bilateral breast reconstruction using abdominal free flaps
A 45-year-old mother of 2 children was diagnosed with left breast cancer, for which a skin-sparing mastectomy (total removal of breast tissue leaving behind the skin envelope) was recommended. Read more
ARTICLES
TL;DR: Breast implants can rupture over time, with signs ...
TL;DR Section: Your age shapes how breast augmentation ...
TL;DR: If diet and exercise haven’t fully shaped your ...
TL;DR: A tummy tuck (abdominoplasty) goes beyond aesthetics ...
Contact Form
1 Orchard Boulevard #10-08 Camden Medical Centre, Singapore 248649
6 Napier Rd, #08-01 Gleneagles Medical Center, Singapore 258499
Tel: +65 6737 4565 | Mobile: +65 8828 4565 | Email: clinic@polarisplasticsurgery.com | Business Hours: Mon - Fri: 9am - 6pm | Sat: 9am - 1pm | Sun/Ph: Closed











